Hydrophobic amino acids and neutral polar amino acids
These amino acids have to be supplied to the body via digested proteins that are then absorbed in the intestine and transported in the blood to where they are needed. The essential amino acids in humans are: histidine, leucine, isoleucine, lysine, methionine, valine, phenylalanine, tyrosine and tryptophan. Essential amino acids are the ones that the body cannot synthesise on its own. There are essential amino acids and non-essential amino acids. These have enzymatic roles and can be used to bind DNA, metals and other naturally occurring ligands. There are 10 non-polar amino acids found in protein core, and there are 10 polar amino acids. Īmino acids can also be characterised as polar or non-polar and these dictate the amino acid function. This prevents sterioisomerism as there aren't four different groups then bonded to the central carbon - there is no chiral centre. The only exception to this being the simplest amino acid glycine with its variable group being another hydrogen atom.

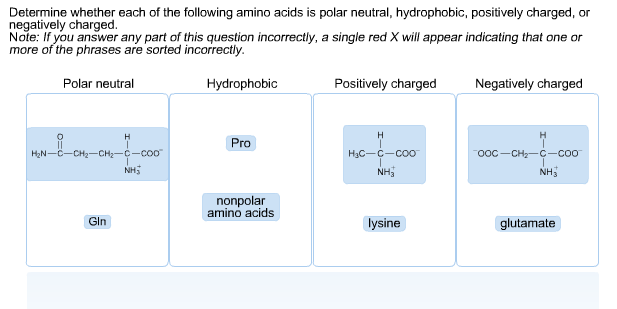
This group varies in the 20 essential amino acids and generally allows amino acids to exhibit sterioisomerism to create optical isomers D and L. All amino acids have an alpha carboxylic acid group, an alpha amine group and a hydrogen atom bonded to a central carbon along with a fourth variable group. When an Amino Acid is part of a protein it is known as an Amino Acid residue, it has the same side chain but it's alpha Amino and carboxyl groups are now part of peptide bonds. Īmino Acids are the monomers that make up proteins by joining in condensation reactions to form peptide bonds between themselves. All amino acids are amphoteric meaning they can act as both a base and an acid due to their amino and carboyxl groups respectively. This bond works as a partial double bond causing the amino acids to have cis/trans isomers. When two amino acids join they form a peptide bond. some bacterial cell walls contain D-isomers. Amino acids exist in proteins as L- optical isomers, however, they can exist as D-isomers in isolated examples, e.g. There are 20 naturally occurring amino acids. To create hydrophilic channels the polar amino acids are generally present in interior pores while nonpolar amino acids are present in regions of surface in contact with the membrane.Amino acids are the building blocks of proteins - they create the proteins primary structure. The stability of the structure is bought by the nonpolar amino acids found in the center of the molecule while the capacity to interact with water molecules is brought about by polar amino acids located on the protein surface. In enzymes, substrate binding to the active site is influenced by hydrophobic and hydrophilic interactions brought about by the location of polar and nonpolar amino acids. The location of polar and nonpolar amino acids depends on the type of protein and its function: Leucine, tryptophan, methionine, proline, valine, alanine, proline and glycine. Therefore, these amino acids are also categorized as hydrophobic. There are no charges present in the alkyl groups. So, these amino acids have neutral charge. This is because these amino acids have equal number of amine groups and carboxylic acids. Non polar amino acids, as the name suggests do not have any charge or polarity. Because of their electronic structure, water particles may acknowledge 2 hydrogen bonds, and give 2, along these lines being all the while occupied with an aggregate of 4 hydrogen bonds. In proteins basically all groups equipped for framing H-bonds (both fundamental chain and side chain, autonomously of whether the buildups are inside an auxiliary design or some other sort of construction) are generally H-attached to one another or to hydrogen atoms. The hydrogen is covalently connected to one of the molecules (called the hydrogen-bond benefactor), yet cooperates electrostatically with the other particle (the hydrogen security acceptor, O).
For a hydrogen attach to be shaped, two electronegative molecules (for instance on account of an alpha-helix the amide N, and the carbonyl O) need to connect with a similar hydrogen.

While hydrophobic amino acids are generally covered inside the center, a more modest division of polar groups are discovered to be covered and charged deposits are presented to dissolvable to a lot more serious level. The favored area of various amino acids in protein particles can be quantitatively described by ascertaining the degree by which an amino acid is covered in the design or presented to solvent. Few examples contains alanine, isoleucine, proline, glycine, valine, leucine, etc. examples of polar amino acids include lysine, serine, and aspartic acid while non-polar amino acids are hydrophobic. Based on polarity they are classified as polar and nonpolar amino acids. Amino acids are classified based on different properties and structures.
